Leading provider of Ketamine infusions.
Welcome to
Actify Neurotherapies
You can also boost your mental health if you decide to pay for homework to get done! Say yes to alternative neurotherapeutic ideas!
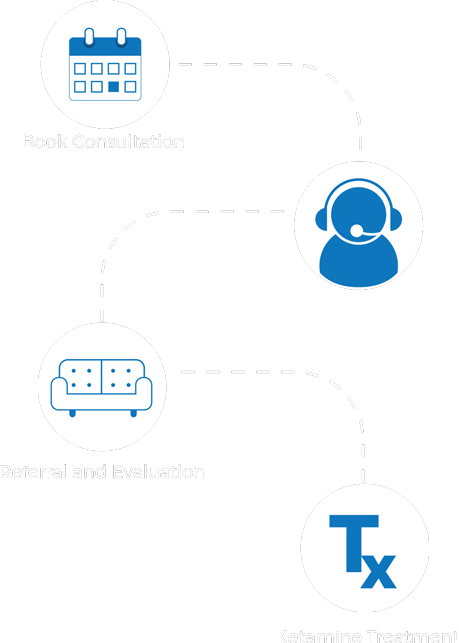
Reserve Your Free 15 min Phone Consultation
Consult with our experienced team to determine if Ketamine is right for you. Call us today or submit this form request for a confidential consultation.
How it Works